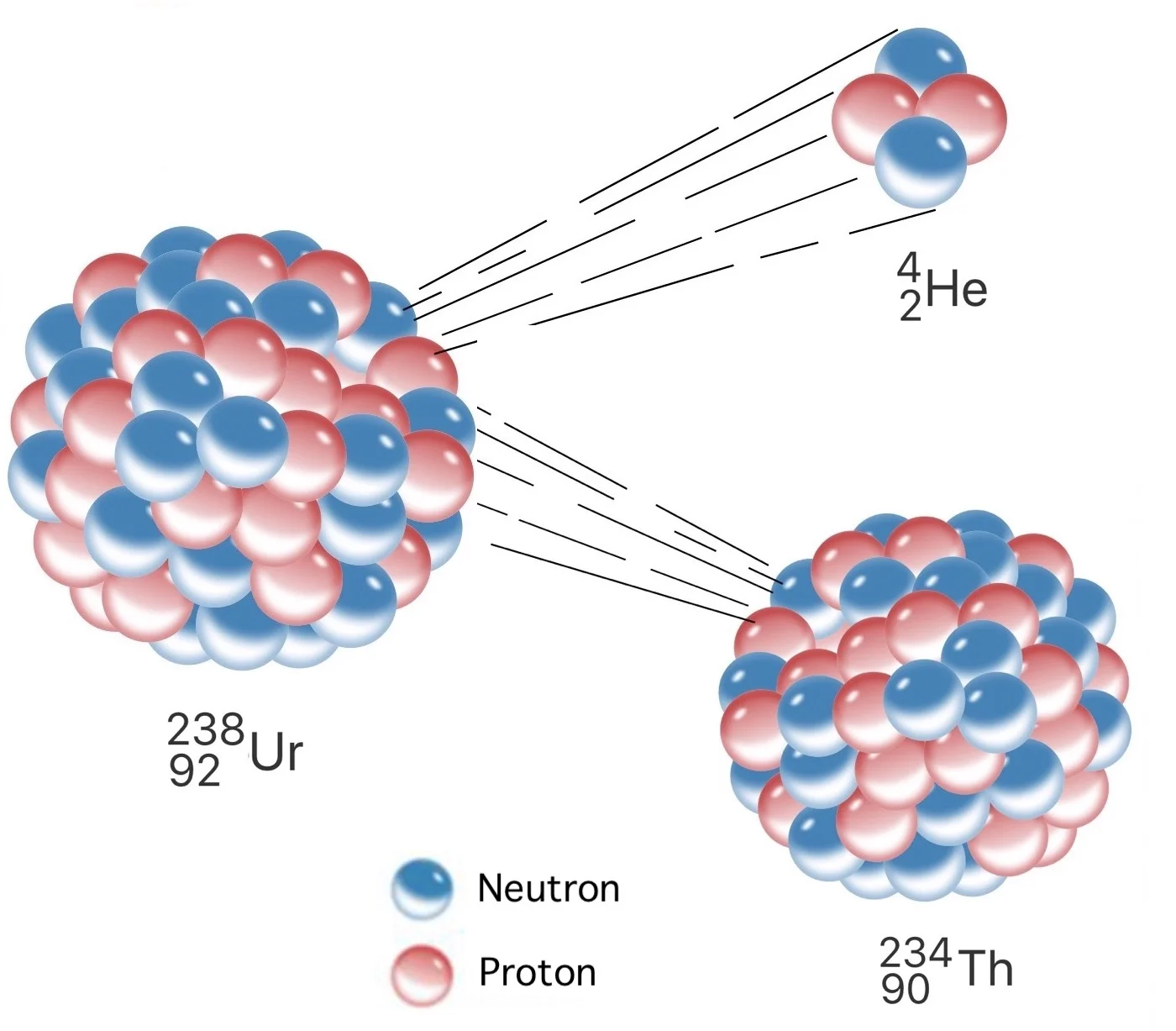  
In fact, many chemists held the view that atoms were the smallest units of matter possible. The modern definition of atomic mass (the weighted average of the atomic masses of all isotopes of an element) was meaningless 150 years ago. Here is where we will begin our journey to periodicity. One such discovery was that of atomic masses. The modern periodic table didn’t spring fully formed from the genius of Mendeleev it was shaped by key discoveries about the elements. So how did Russian chemist Dmitri Ivanovich Mendeleev and the other creators of the periodic table (arguably six of them) bring order to the elements? How did they create a tool that would ultimately house 118 elements when they knew only 62 of them? And why does Mendeleev get all the credit? Mass + reactions = periodic table It even predates knowledge of the noble gases. However, the periodic table predates knowledge of atomic numbers and subatomic particles (yes, including electrons). If you know about atomic numbers and electron shells, recreating the periodic table is simple. You would order them by increasing atomic number and create a new row when you hit a noble gas. If I gave you all of the elements on cards and told you to recreate the periodic table, you probably wouldn’t have much trouble. You can compare electronegativity, ionization energy, atomic radius, chemical reactivity, and more. You can determine the electron configuration of any atom, simply from its place. The periodic table is an elegant demonstration of properties of elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
